Key Features & Functionality
01. Participant Enrollment & Study Tracking
Track participants from initial screening through study completion. Coordinators can register new participants, assign them to specific studies and sites, and update enrollment statuses, such as screened, enrolled, withdrawn, or completed. Consent status and enrollment dates remain attached to each participant profile, giving research teams a clear view of study participation at any moment.
02. Study Visit Scheduling & Monitoring
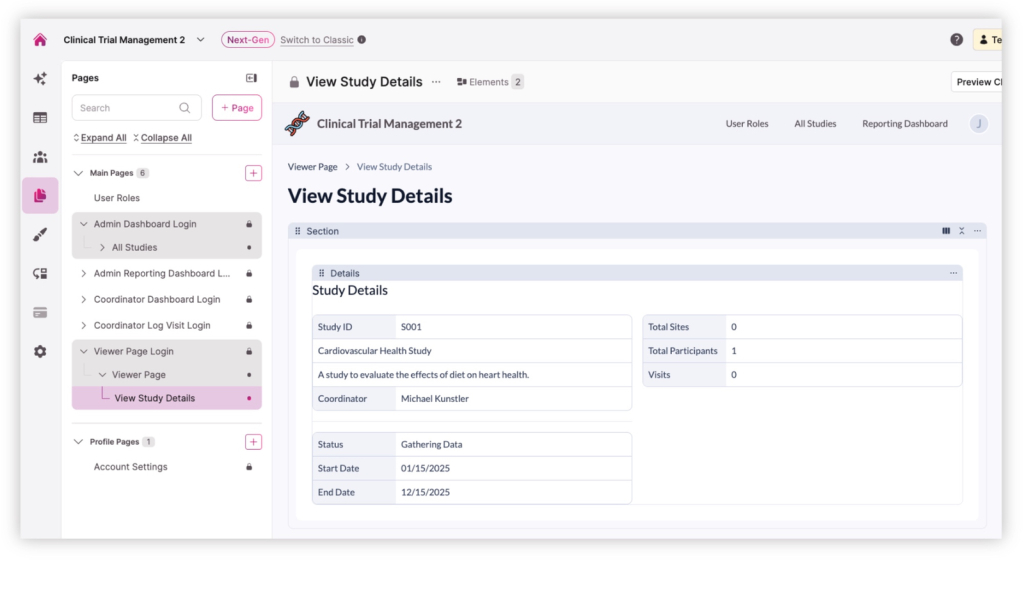
Plan and manage participant visits across multiple studies and sites. Coordinators can schedule visits, track whether they were completed or missed, and update visit details directly within the system. Each visit record connects to the participant and study, allowing research teams to easily monitor adherence to study protocols and timelines.
03. Flexible Data Collection Forms
Attach customizable data collection forms to each visit. Research teams can capture vitals, surveys, laboratory results, and other study-specific data using simple form-based workflows. Fields can be adjusted as protocols evolve, and multiple forms can be completed for each visit to support different types of clinical observations and research data.
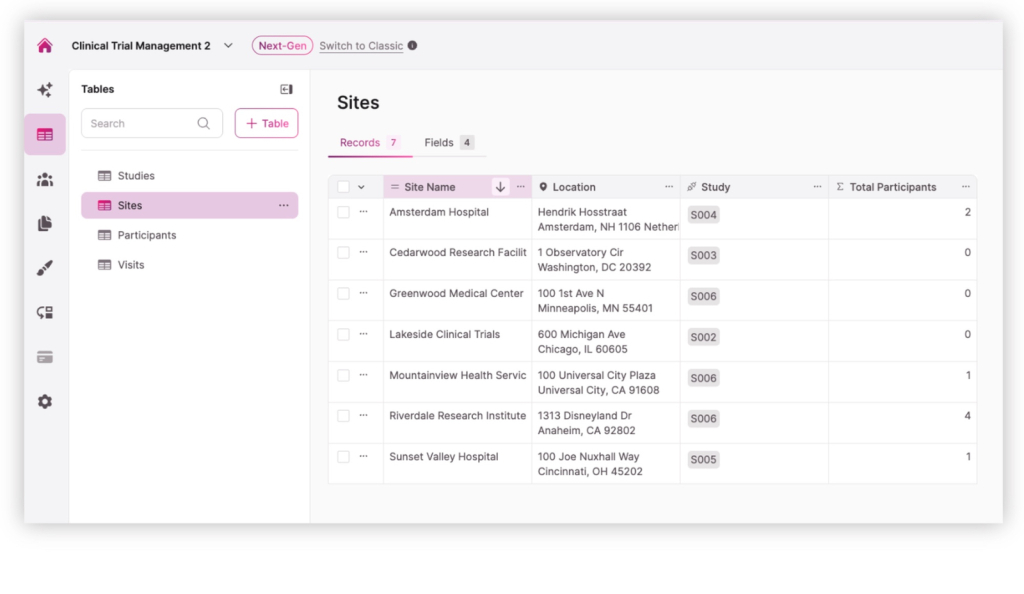
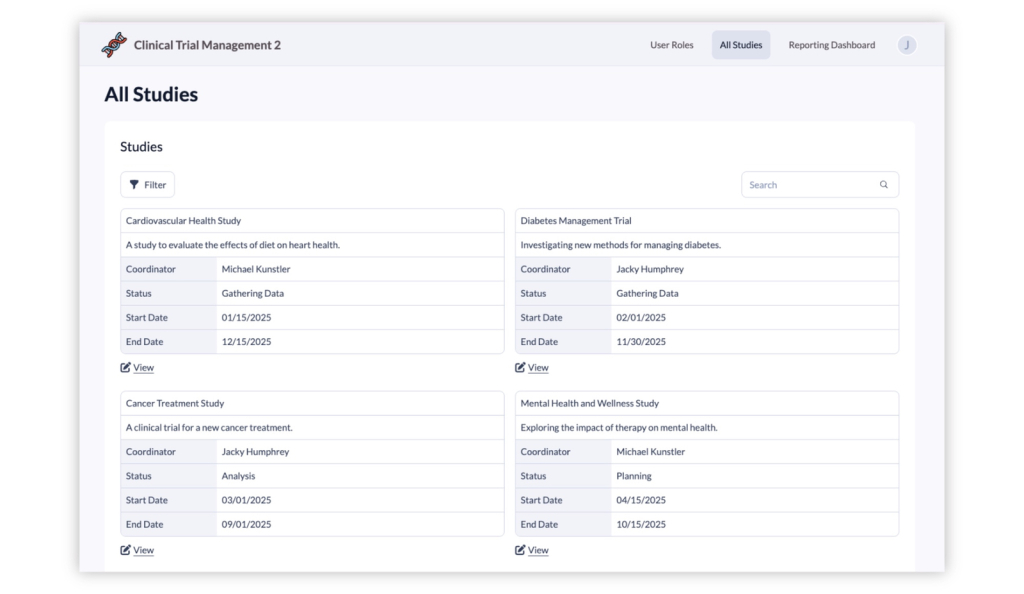
04. Multi-Site Study Management
Manage studies across multiple research sites from a single application. Administrators can create studies, add participating locations, and assign coordinators or staff to the appropriate site. This structure helps organizations coordinate research activity across hospitals, clinics, or partner institutions while keeping study data centralized and organized.
05. Role-Based Access for Research Teams
Provide separate portals for administrators, coordinators, and viewers. Administrators can oversee studies and site assignments, coordinators manage participant enrollment and visits, and viewers can review study progress or reports. Role-based permissions ensure that users only see the data and functions relevant to their responsibilities.
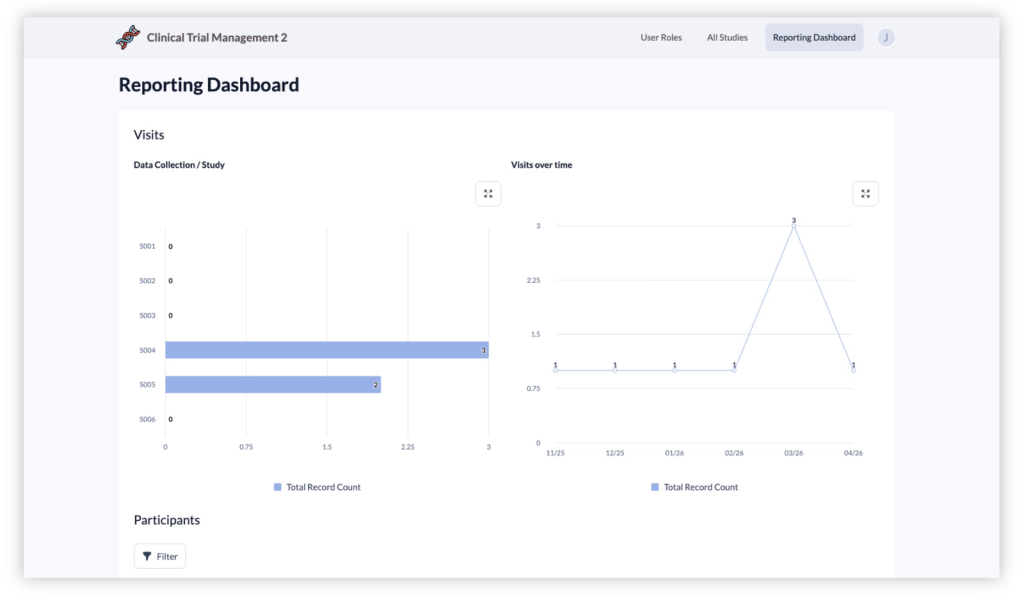
06. Study Progress Reporting & Dashboards
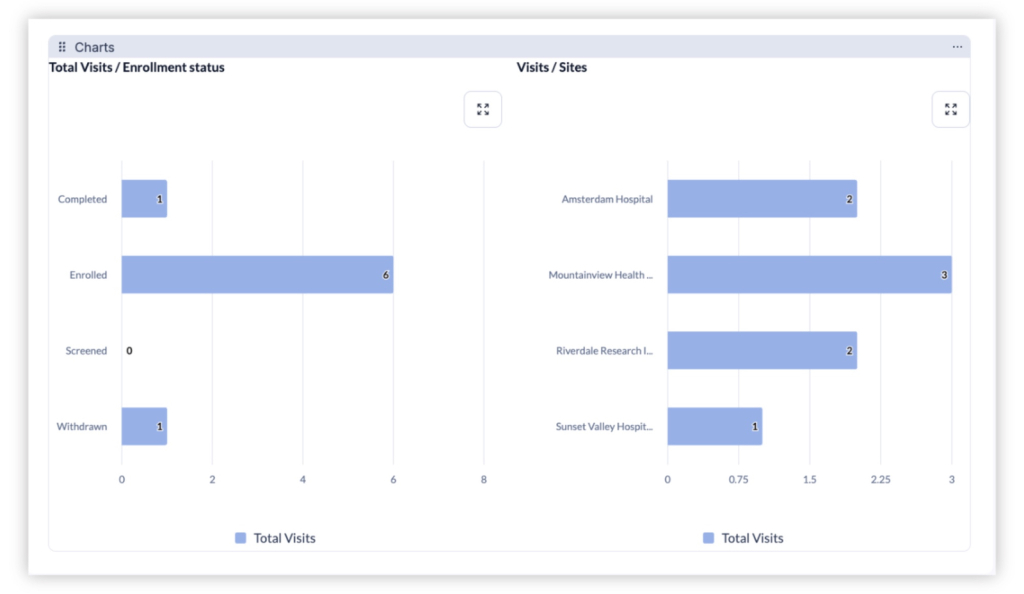
Monitor clinical trial progress with built-in dashboards and reports. Teams can view enrollment counts by study or site, track visit completion rates, and review participant statuses in real time. These insights help research leaders identify bottlenecks, ensure protocol adherence, and keep trials moving forward efficiently.
Use Cases for the Clinical Trial Template
Participant Enrollment Tracking
Track participants from screening to completion with a centralized participant registry. Coordinators can quickly review enrollment status, consent records, and site assignments without relying on spreadsheets or disconnected tools.
Clinical Visit Coordination
Manage participant visit schedules and monitor completion across study timelines. Coordinators can track scheduled, completed, and missed visits while maintaining a clear record of participant interactions.
Multi-Site Trial Management
Support research studies conducted across multiple hospitals, clinics, or research centers. Assign coordinators and staff to specific sites while maintaining centralized visibility into study performance.
Structured Research Data Collection
Capture study data using standardized forms linked to participant visits. Teams can record vitals, surveys, lab information, and other study measurements in a consistent format that supports accurate reporting and analysis.
Study Oversight for Research Administrators
Provide research leadership with dashboards showing enrollment progress, visit completion, and participant status across studies and sites. This visibility helps ensure studies remain on track and compliant with protocols.
Clinical Research Operations Hub
Replace scattered spreadsheets, shared drives, and manual tracking systems with a single structured platform for participant management, study coordination, and research reporting.
Traditional Tools vs. Knack Health
Knack wins over traditional work order tools, docs, and spreadsheets.
| Traditional Tools | Knack Health |
|---|---|
| Fragmented Trial Tracking: Clinical trials are often managed through spreadsheets, emails, and disconnected systems, making it difficult to track participant progress or study performance. | Centralized Study Management: Knack brings participant records, study data, visit tracking, and site coordination into one structured platform designed for clinical research workflows. |
| Manual Coordination: Coordinators must manually track visits, enrollment statuses, and site assignments, which increases administrative burden and the risk of errors. | Automated Workflows: Trigger notifications for scheduled visits, track enrollment milestones, and support coordinators with automated processes that reduce manual work. |
| Limited Data Visibility: Research leaders often lack real-time insights into enrollment progress, visit completion rates, or site performance. | Real-Time Research Visibility: Dashboards and reports provide instant insights into participant enrollment, visit completion, and study progress across sites. |
| Inconsistent Data Collection: Paper forms and ad-hoc data entry can lead to inconsistent study data and difficulty maintaining standardized records across sites. | Secure, Role-Based Access: Teams can control who sees and edits data through configurable permissions, encrypted storage, and HIPAA-ready hosting options with a BAA available. |